|
12/22/2023 0 Comments 4 blocks of periodic table Types of Blocks present in Periodic Table: The ones that are marked with the red color border s-block. They are arranged in a tabular format with rows representing periods and columns representing groups. The periodic table shown above demonstrates how the configuration of each element. You can effortlessly find every single detail about the elements from this single Interactive Periodic table. Periodic Table: The elements in the periodic table are arranged according to their rising atomic number and recurrent chemical characteristics. So Oxygens electron configuration would be O 1s22s22p4. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Let me know in the comments, has this article helped you or not? Free Gift for you: Interactive Periodic Table I hope you have completely understood the arrangement of Periodic table. There are 4 blocks on the Periodic table which describes the position of valence electrons in the s, p, d or f orbitals. Later on, we saw that there are groups as well as periods on the Periodic table. Hence for grouping the elements according to the similar chemical properties, the elements of the Periodic table are arranged in the increasing order of their atomic number. These electrons are responsible for the chemical properties of an element. In the very beginning of this article, we have seen that the elements of periodic table are arranged in the increasing order of their atomic number.Īnd for a neutral atom, the number of protons are equal to the number of electrons. Similarly, elements which are in the p block have the valence electrons in p-orbitals.Įlements in d block have valence electrons in d-orbitals and elements of f block have valence electrons in f-orbitals. The elements which are in the s block have the valence electrons in s-orbitals. Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. 
There are 4 blocks in the Periodic table: s block, p block, d block and f block. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. The elements of the Periodic table are also classified block wise. The first element having the least atomic number 1 (i.e hydrogen) is placed on the first cell of the Periodic table. So now you know that the elements are arranged according to the increasing order of their atomic number. Interactive periodic table with up-to-date element property data collected from authoritative sources. 
This new spectrum shows the peak frequencies near 2 Hz, 4 Hz, and 6. Step by step arrangement (from 1 st to 118 th element) The reason is that the discrete Fourier transform of a time-domain signal has a periodic. Hence the Periodic table is organized on the basis of Atomic number. In other words, Atomic number indicates the number of electrons, which are responsible for the chemical properties of elements. Hence to classify the elements on the basis of similarities in their chemical properties, they are arranged in the Periodic table on the basis of atomic number. 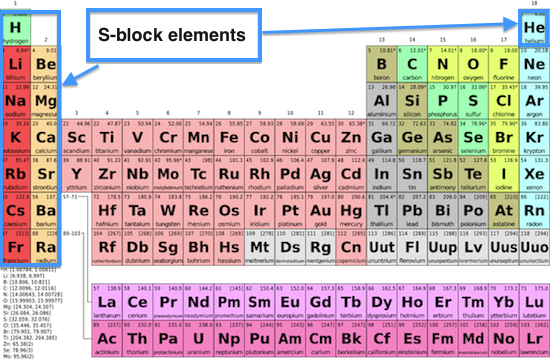
The electrons present in the outermost orbit represent the chemical properties of the elements. So far in the above discussion, we have seen that the atomic number represents the number of electrons of an element. Number of protons (or atomic number) = Number of electrons Summary (Why is Periodic table arranged by atomic number?) The \(4f\) sublevel belongs to the sixth period.Here you can see that the helium atom has 2 protons and the number of electrons are also 2. 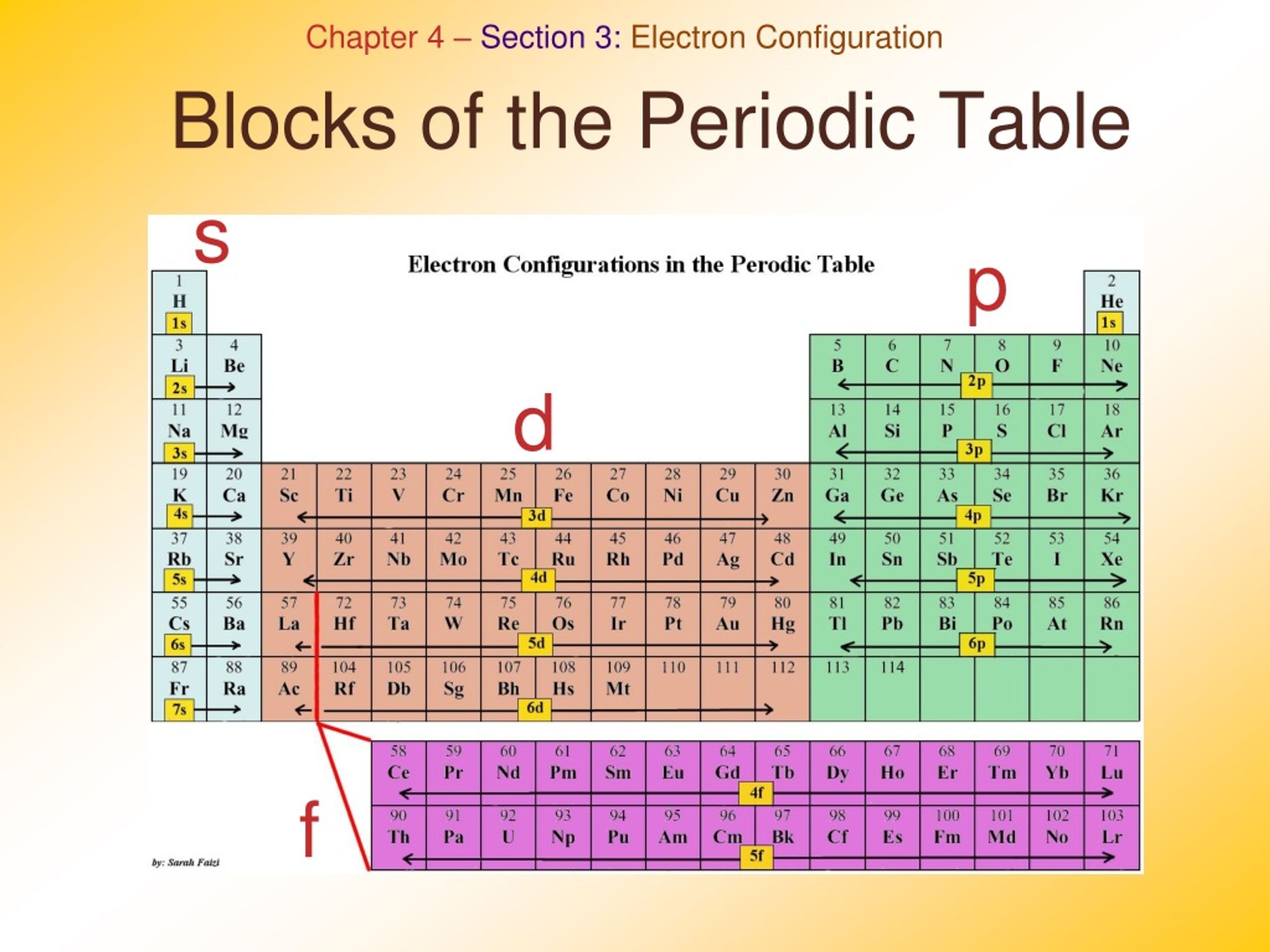
The \(f\) sublevel is always two levels behind. In other words, the \(3d\) sublevel fills during the fourth period. The figure also illustrates how the \(d\) sublevel is always one principal level behind the period in which that sublevel occurs. The modern periodic table is divided into how many. \) (Credit: Christopher Auyeung and Joy Sheng Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window)) They are : (i) s-block, (ii) p-block (iii) d- block and (iv) f-block. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
